Updated: PETAH TIKVA, Israel, May 6, 2026, 14:59 (IDT)
- Zebra Medical Vision is now Nanox.AI, the medical imaging software arm of Nasdaq-listed Nanox.
- Radiology AI has moved into a crowded regulated market, with hospitals, payers and watchdogs asking for proof that tools change care.
- Nanox has new commercial openings, but its filings show losses and financing risk.
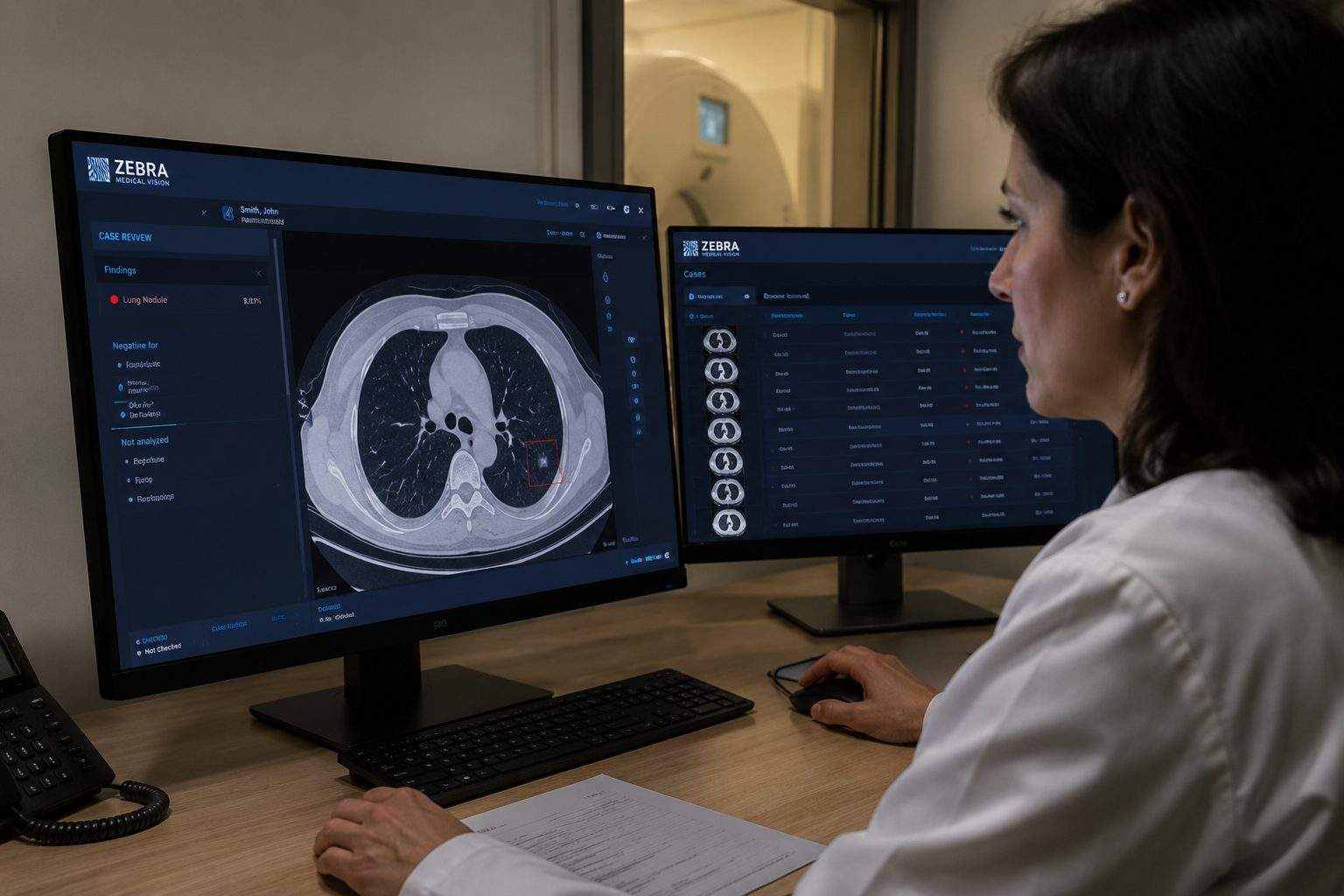
Nanox is trying to turn Zebra Medical Vision’s scan-reading software into a hospital business, not just an AI demo. The company said it had accelerated work around Nanox.AI, added Cedars-Sinai as a clinical-trial partner for an aortic valve calcification tool and pushed U.S. deployment of Nanox.ARC systems, with Chief Executive Erez Meltzer calling the fourth quarter “strong momentum.” Nano-X Imaging LTD.
The timing matters because medical imaging AI is now a regulated market, not a side project. The U.S. Food and Drug Administration says its AI-enabled device list identifies devices authorized for U.S. marketing, while an industry analysis of FDA data counted 1,451 AI-enabled authorizations through 2025, including 1,104 in radiology.
Payers are also asking for harder proof. NICE, the UK health-technology guidance body, said five AI tools, including Nanox AI’s HealthVCF and HealthOST, can be used in the NHS during a three-year evidence-generation period and paid for with core NHS funding; a vertebral fragility fracture, or VFF, is a spine fracture often linked to weak bones from osteoporosis.
Zebra Medical Vision is no longer independent. Nanox’s 2026 annual filing said the company bought 100% of Zebra in November 2021 and that Zebra, now called Nanox AI, was described as having eight FDA-cleared and 11 CE-marked AI imaging solutions; the filing put the basic purchase price at $100 million in Nanox shares, with other consideration tied to costs and milestones.
The old Zebra pitch was to mine scans already sitting inside hospitals. Nanox told investors that Nanox AI develops machine-learning platforms, meaning software trained on large data sets to recognize patterns, based on a database of more than 500 million imaging scans, and sells AI imaging tools to hospitals, health plans, imaging companies, pharmaceutical firms and insurers.
One cardiac product, HealthCCSng, reviews routine adult CT scans for coronary artery calcium, or CAC, the calcified plaque in heart arteries that can signal coronary artery disease. FDA materials say the tool is not for stand-alone clinical decision-making and that the final diagnosis remains with the clinician; the scan type is non-gated and non-contrast, meaning it is not synchronized to the heartbeat and does not use contrast dye.
Nanox has also pushed beyond heart and bone. In 2024, it received FDA 510(k) clearance for HealthFLD, software that analyzes liver attenuation on routine CT scans in adults aged 18 to 75 to support detection of fatty liver linked to metabolic dysfunction-associated steatotic liver disease, or MASLD, the newer name for a common fatty-liver condition tied to metabolic risk.
The strongest clinical narrative around Nanox.AI remains bone health. Oxford’s NDORMS said early ADOPT study findings showed HealthVCF identified up to six times more patients with vertebral compression fracture than the UK NHS national average and flagged more than 2,400 previously unknown patients for follow-up; Professor Kassim Javaid said the data showed “the potential of an AI-directed pathway,” while Nanox Chief Medical Officer Orit Wimpfheimer pointed to possible “cost-savings” from earlier detection. NDORMS
Competition is not thin. NICE’s same assessment listed Aidoc Medical’s BriefCase-Triage, Avicenna.AI’s CINA-VCF Quantix and IB Lab FLAMINGO alongside Nanox’s two CT-based bone tools, and Axios reported in April that Aidoc raised $150 million in Series E funding, underscoring how much capital is still chasing clinical AI in radiology.
But clearance and clinical promise do not remove the risk. Reuters reported in February that regulators had received injury and malfunction reports involving AI-enabled devices and quoted Dr. Alexander Everhart of Washington University as saying the FDA’s traditional device approach was “not up to the task”; NICE separately flagged false positives, radiology workload, DEXA scan referrals and unequal performance across patient groups as issues to monitor. Reuters
Nanox’s own numbers show the commercial climb. Its annual filing showed 2025 revenue of $13.0 million, including $1.0 million from AI, against a net loss of $75.0 million and $40.8 million of cash used in operations; management also said there was substantial doubt about its ability to continue as a going concern under its current operating plan.
That leaves a narrower story than the simple claim that AI can read and diagnose medical images. A 510(k) clearance shows a device is substantially equivalent to a legally marketed device and can be marketed in the United States, but it does not settle whether hospitals will buy the workflow, whether payers will keep funding it, or whether the software changes care enough to matter.